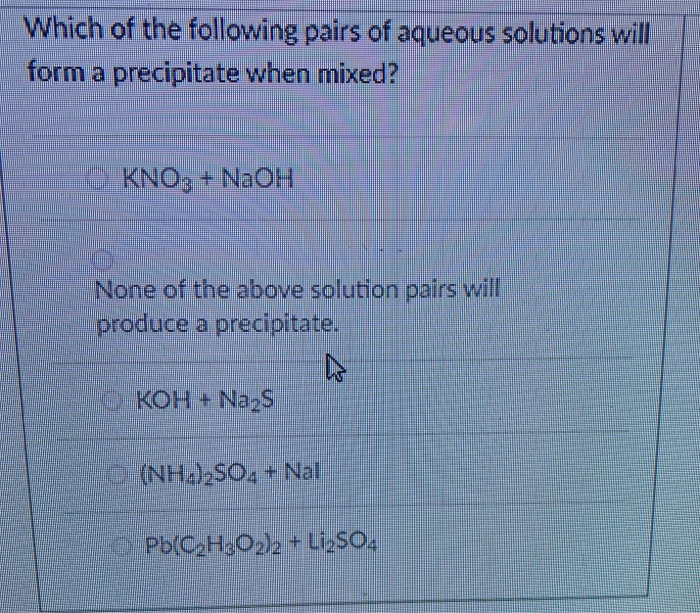
There are many other factors that can affect solubility, but these rules are a good first step to determine the outcome of aqueous solution reactions. The solubility rules are a useful guideline to predict whether a compound will dissolve or form a precipitate. The finished reaction is:Ģ KCl(aq) Pb(NO 3) 2(aq) → 2 KNO 3(aq) PbCl 2(s) This means PbCl 2 is insoluble and form a precipitate. Chlorides are soluble in water with the exception of silver, lead and mercury. and as a solvent in The addition of GBL to an aqueous solution of 3. KNO 3 will remain in solution since all nitrates are soluble in water. In acidic water, a mixture of the lactone and acid forms coexist in an equilibrium. The products should rearrange the ions to: What would be the expected products and will a precipitate form? The resulting balanced reaction would be:Ģ AgNO 3(aq) MgBr 2 → 2 AgBr(s) Mg(NO 3) 2(aq) The other compound Mg(NO 3) 2 will remain in solution because all nitrates, (NO 3) -, are soluble in water. Are the products soluble in water?Īccording to the solubility rules, all silver salts are insoluble in water with the exception of silver nitrate, silver acetate and silver sulfate. The state of the products needs to be determined. The balanced reaction would be:Ģ AgNO 3(aq) MgBr 2 → 2 AgBr(?) Mg(NO 3) 2(?) For example, a silver nitrate solution (AgNO 3) is mixed with a solution of magnesium bromide (MgBr 2). The question remains, will AD or CB remain in solution or form a solid precipitate?Ī precipitate will form if the resulting compound is insoluble in water. This reaction is generally a double replacement reaction in the form: When two aqueous solutions are mixed, the ions interact to form products. These solutions are represented in chemical equations in the form: AB(aq) where A is the cation and B is the anion. This guide will show how to use the solubility rules for inorganic compounds to predict whether or not the product will remain in solution or form a precipitate.Īqueous solutions of ionic compounds are comprised of the ions making up the compound dissociated in water. Iron alkoxide complexes are formed when ferric chloride reacts with alkali metal alkoxides.When two aqueous solutions of ionic compounds are mixed together, the resulting reaction may produce a solid precipitate.Using CuCl2 will form CucO3 which is insoluble in water. 
\(FeCl_3(s) NH_4OH(aq)\rightarrow Fe(OH)_3(s) NH_4Cl\) Which solution forms a precipitate when mixed with a solution of aqueous Na2CO3 The answer is CuCl2 however I am unclear how that was gotten. A reddish-brown precipitate of ferric hydroxide is formed when ferric chloride and ammonium hydroxide interact.Ferguson Senior High Scholl Course Title CHE INORGANIC Uploaded By mosesbazzi. When aqueous solutions of are mixed a precipitate. the formed sodium chloride or bromide precipitates out from the solution. When aqueous solutions of are mixed a precipitate forms A KOH and BaNO 3 2 B Li. A reddish-brown precipitate is produced when sodium hydroxide is added to a solution of ferric chloride. Place all aqueous solutions and extracts in the sodium hydrogen carbonate waste.In the structure of ferric chloride, which frequently combines iron and chlorine, iron has a 3 oxidation potential. Ferric chloride is moderately soluble in non-polar solvents and very soluble in liquids with donor properties. When wet it is corrosive to aluminium and most metals. precipitate forms when aqueous solutions and are mixed: If a precipitate will. Aluminium and most metals are corroded by it when moist. Question: Does a reaction occur when aqueous solutions of zinc nitrate and. To predict whether a precipitate forms when we mix aqueous solutions of two strong. 1 that solutions are defined as homogeneous mixtures that are mixed so thoroughly that. Analyze The two salts are in solution, so they both must dissociate in. We then need to write the net ionic equation and compare it with the one from the previous Sample Exercise. 
Using the names of the reactants, we are asked to identify the precipitate. It is a brown-black solid that doesn’t burn. A precipitate is an insoluble solid formed by a reaction in solution. Water forms hydrogen bonds with ions and other polar substances. Collect and Organize Mixing two solutions causes an insoluble compound to precipitate. Ferric chloride is normally formed when iron and chlorine interact. 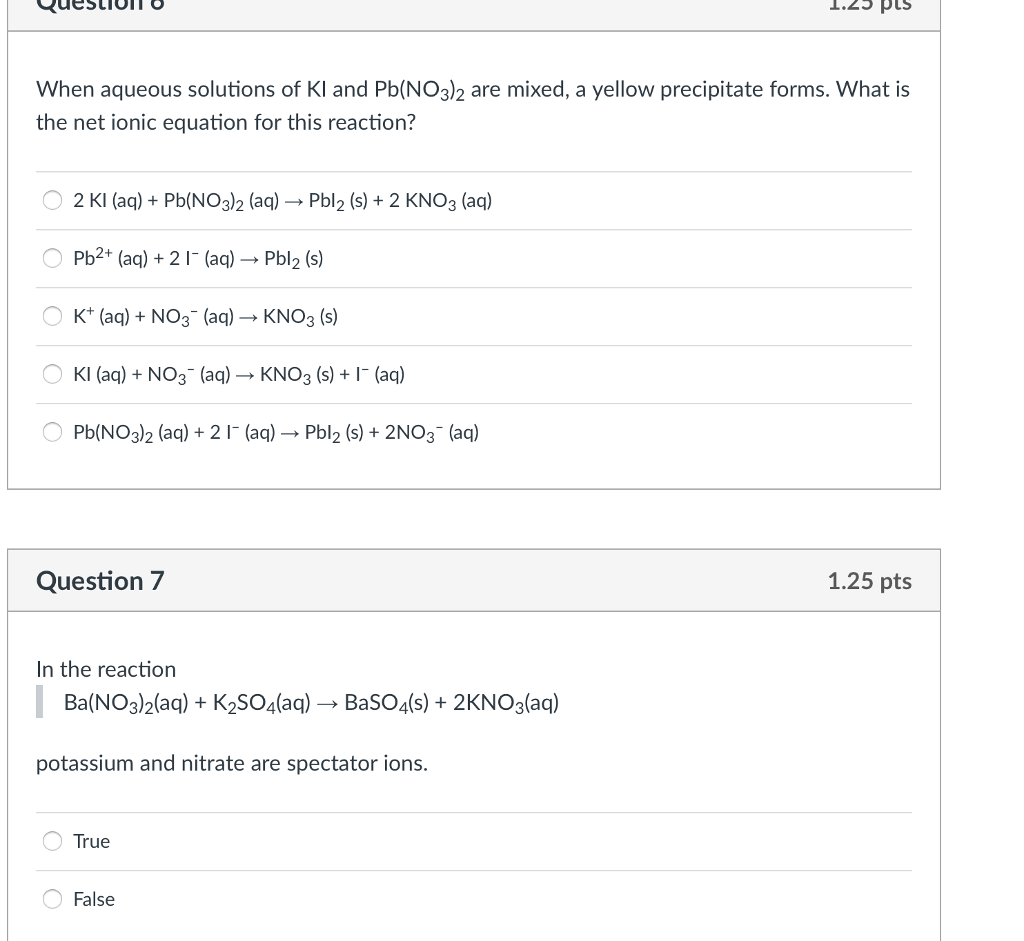
Ferric Chlorideįerric chloride is an inorganic compound. In this article, we will look into its structure, formula, properties and uses in detail.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
