HRTEM/EDS confirms the expected core-shell gold-coated magnetic nanoparticle morphology ( Fig. 6 displays HRTEM images of mono-disperse Fe 3O 4-AuNPs. 
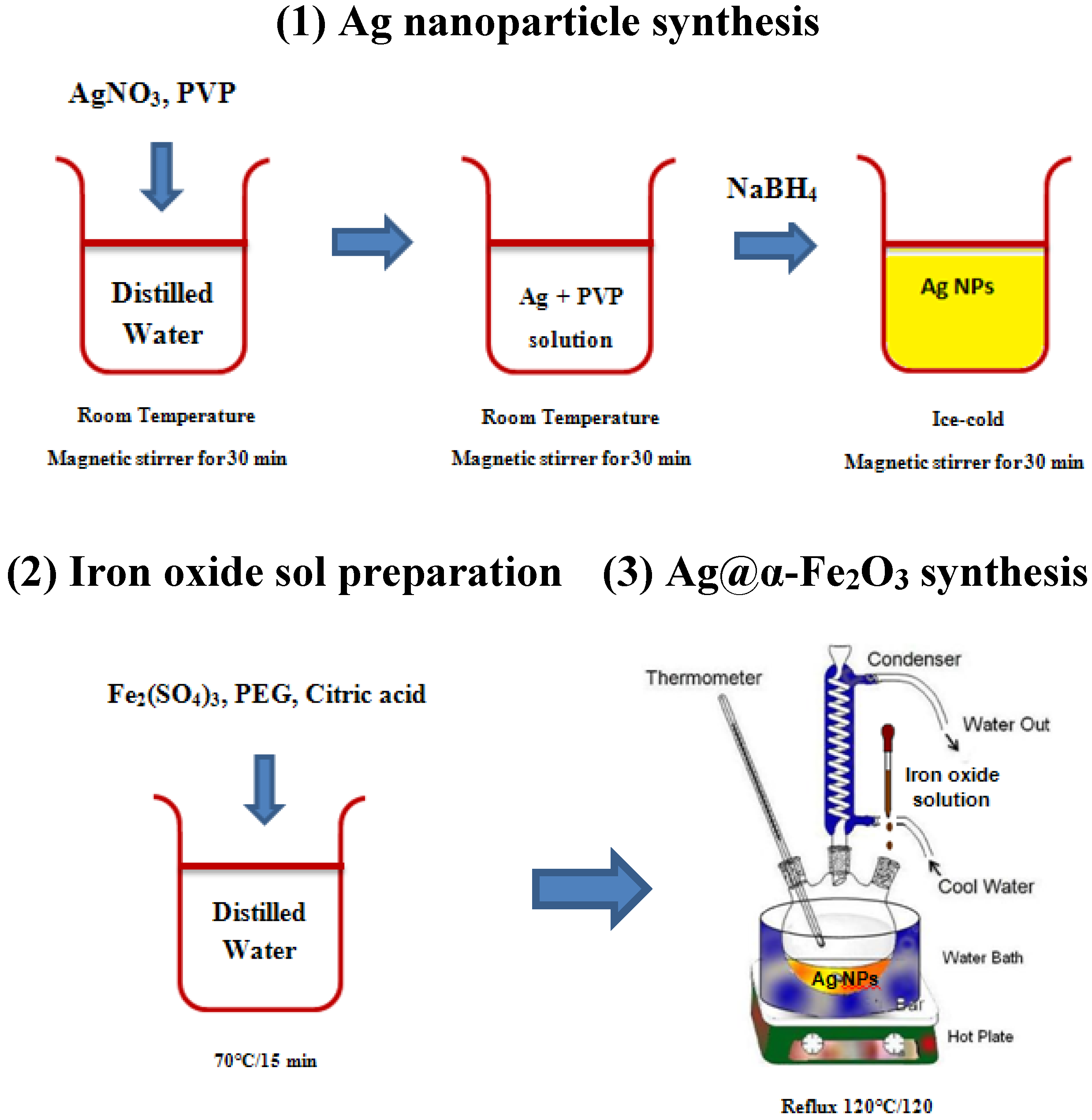
Although there still remained a notable decrease in yield, transfer of the Fe 3O 4-AuNPs with DMAP was found to be successful. The transferred DMAP-capped Fe 3O 4-AuNPs were characterized by HRTEM/EDS and UV–vis absorption spectroscopy. After 1 h with vigorous stirring, it was found that most of the Fe 3O 4- AuNPs were transferred and soluble in water. Adding a 0.5 M solution of DMAP in water results in the Fe 3O 4-AuNPs spontaneously transferring across the phase boundary into the water layer. Before transfer, the Fe 3O 4-AuNPs capped with oleylamine/oleic acid are soluble in hexanes (top layer). 5 displays the location of the Fe 3O 4-AuNPs over time during transfer. The scale of the image was reset from metres to pixels. Transmission electron micrograph images were analyzed for nanoparticle size distribution using ImageJ software. EDS of each Fe 3O 4-AuNPs sample was taken at a 20° take off angle with an Oxford X-ma × 80 mm EDS detector using Aztec software. Images were taken at 1–2 μm, 100–200 nm and 5–10 nm for each grid. The TEM grids were allowed to dry for 4–24 h depending on the solvent. Grids were prepared by placing 4 μL of Fe 3O 4-AuNP (in various solvents) on a TEM grid. All images were taken on dry 300 mesh × 83 μm pitch carbon coated copper TEM grids at room temperature. Transmission electron micrographs were taken with a FEI Technai G2 F20 TEM at the Carleton University Nano-imaging Facility, with a field emission source at a voltage of 200 kV using Gatan Microscopy Suite 2 V. Fe 3O 4-AuNPs were prepared as described and analyzed at each step during synthesis. The UV–vis absorption characterization of the Fe 3O 4-AuNPs was performed using a Cary 300 Bio UV–vis spectrophotometer (Varian, Santa Clara CA). Transfer of Fe 3O 4-AuNPs with 4-dimethyl(amino)pyridine Solutions appeared dark red-purple in colour, and were stored in glass covered by foil at room temperature. This procedure was repeated multiple times to obtain purified Fe 3O 4-AuNPs in hexanes. The nanoparticles were then redispersed in a solution of 10 mL of hexanes, 0.25 mL of oleic acid and 0.25 mL of oleylamine. The supernatant solution was decanted as waste and the precipitated nanoparticles were washed three times with 15 mL of absolute ethanol. The flask was then placed on a magnet for 5–10 min to magnetically separate the particles from the supernatant solution. The solution was agitated gently resulting in a visible aggregation of the nanoparticles and a colour change from dark purple to dark blue/black. 5 mL of the solution was transferred to a 125 mL Erlenmeyer flask and 15 mL of ethanol was added to it. The solution was allowed to cool to room temperature under argon overnight. This suspension was heated slowly (10 ☌/ min) until 180–190 ☌ and maintained at this temperature under reflux for 3 h. To this solution, 0.83 g of Au(ac) 3, 3.1 g of 1,2-hexadecanediol, 0.5 mL of oleic acid and 3 mL of oleylamine were added quickly. 10 mL of previously prepared Fe 3O 4 in phenyl ether (above reaction product) was added to 30 mL of phenyl ether with vigorous stirring.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
